The following document provides introductory information on metabolites and the field of metabolomics.
Metabolism is the set of life-sustaining, enzyme-catalyzed chemical transformations within the cells of living organisms. These reactions allow organisms to perform necessary operations such as growth and reproduction, structural maintenance, digestion, and environmental response. Intermediary or intermediate metabolism is the transformation of nutrients into cell components.
Metabolism is usually divided into two categories: catabolism, which breaks down organic matter and harvests energy by way of cellular respiration; and anabolism, which uses energy to construct the components of cells such as proteins and nucleic acids
Catabolism
Catabolism is the set of metabolic pathways that breaks down large molecules, such as polysaccharides, lipids, nucleic acids and proteins into smaller units, monosaccharides, fatty acids, nucleotides, and amino acids, respectively, to release energy. Catabolism therefore provides the chemical energy necessary for the maintenance and growth of cells.
As cells continue to break down the newly available monomers, their degradation results in a mix of waste products, heat, and chemical free energy, which is used to drive the synthesis of adenosine triphosphate (ATP). ATP assists in the transfer of energy released by catabolism to the anabolic reactions that require energy.
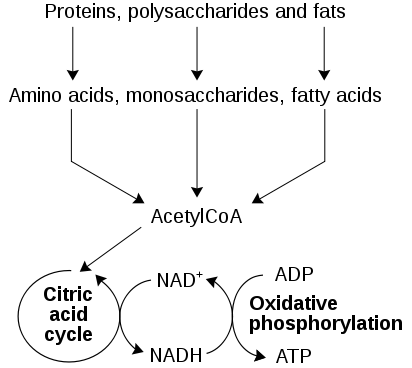
Figure 1. Process of catabolism
Most of the known signals that control catabolism are hormones, though many are also the molecules involved in metabolism itself.
Anabolism
Anabolism is the set of metabolic pathways that construct molecules from smaller units. Catabolism is often understood as destructive metabolism while anabolism is constructive metabolism. Many anabolic processes are powered by the hydrolysis of adenosine triphosphate (ATP), a byproduct of some catabolic processes.
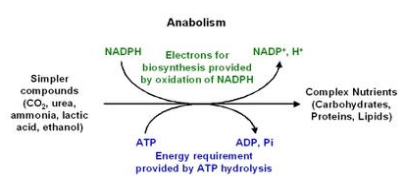
Figure 2. Process of anabolism
Anabolic processes tend toward "building up" organs and tissues. These processes produce growth and differentiation of cells and increase in body size, a process that involves synthesis of complex molecules. Examples of anabolic processes include the growth and mineralization of bone and increases in muscle mass.
Endocrinologists have traditionally classified hormones as anabolic or catabolic, depending on which part of metabolism they stimulate. The classic anabolic hormones are the anabolic steroids, which stimulate protein synthesis and muscle growth, and insulin.
Metabolites, the intermediates and products of metabolism, affect enzymatic activity (sometimes as a cofactor), defend the cell, and promote extra-organismal interaction.
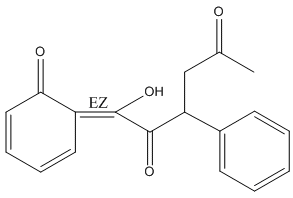
Figure 3. Structure of Warfarin metabolite
The concentration and the properties of the enzymes determine the quantity of metabolites. Therefore, their level of abundance is a complex function of many different regulatory processes inside the cell. The number of metabolites represents integrative information of the cellular function, defining the phenotype of a cell or tissue in response to genetic or environmental changes
Examples of various cellular processes that determine metabolite abundance include:
- regulation of transcription and translation
- regulation of protein–protein interactions
- allosteric regulation of enzymes through their interaction with metabolites
Metabolic Pathway
In biochemistry, metabolic pathways are series of chemical reactions occurring within a cell. In each pathway, a principal chemical is modified by a series of chemical reactions. Enzymes catalyze these reactions, and often require dietary minerals, vitamins, and other cofactors in order to function properly. Because of the many chemicals (a.k.a. metabolites) that may be involved, metabolic pathways can be quite elaborate. In addition, numerous distinct pathways co-exist within a cell. This collection of pathways is called the metabolic network. Pathways are important to the maintenance of homeostasis within an organism. Catabolic (break-down) and Anabolic (synthesis) pathways often work interdependently to create new biomolecules as the final end products.
A metabolic pathway involves the step-by-step modification of an initial molecule to form another product. The resulting product can be used in one of three ways:
- To be used immediately,
- To initiate another metabolic pathway, called a flux generating step
- To be stored by the cell
A molecule called a substrate enters a metabolic pathway depending on the needs of the cell and the availability of the substrate. An increase in concentration of anabolic and catabolic intermediates and/or end-products may influence the metabolic rate for that particular pathway.
A team of scientists at Georgia Tech University mapped the complex series of human metabolic pathways in one huge poster. WARNING: VERY high resolution image.
Metabolome
The metabolome, typically defined as the collection of small molecules produced by cells, offers a window for interrogating how mechanistic biochemistry relates to cellular phenotype. With developments in mass spectrometry, it is now possible to rapidly measure thousands of metabolites simultaneously from only minimal amounts of sample.
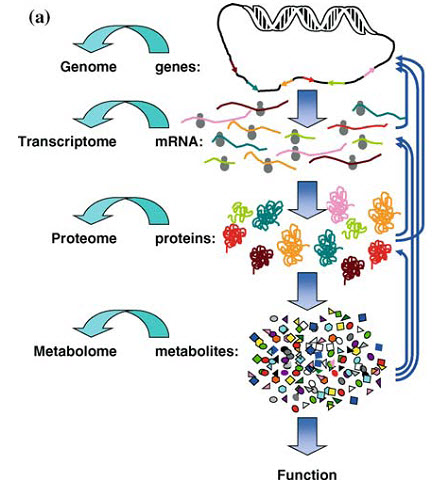
Figure 4. The metabolome, the collection of metabolites and cellular byproducts, determine function
Unlike the proteome, for example, the metabolome consists of extremely diverse chemical compounds from ionic inorganic species to hydrophilic carbohydrates, volatile alcohols and ketones, amino and non-amino organic acids, hydrophobic lipids, and complex natural products. That complexity makes it virtually impossible to simultaneously determine the complete metabolome.
Metabolomics is the systematic study of the unique fingerprints and byproducts of specific, life-sustaining chemical transformations in the cells of living organisms. Using metabolites, metabolomics researchers study the various organisms’ small-molecule profiles that suggest specific biological processes.
Figure 5. Cells engage in metabolism
There are two types of metabolomic analysis: targeted and untargeted metabolomics.
Targeted Metabolomics
Targeted metabolomics selects a number of specific metabolites to analyze, using their known set of characteristics to monitor their levels across an experiment. Targeted analytical techniques have been in use for centuries, allowing researchers to build increasingly more sophisticated methods of detection. For instance, triple quadrupole mass spectrometry, known as QqQ, can perform selected reaction monitoring experiments.
This has led to scientists creating routine methods for analyzing most of the metabolites in central carbon metabolism, as well as amino acids and nucleotides at their naturally occurring physiological concentrations.
These techniques are highly sensitive, quantitatively reliable, and robust enough to measure a significant number of biologically important metabolites with relatively high throughput.

Figure 6. A mass spectrometer
Untargeted Metabolomics
Untargeted metabolomics casts a much more indiscriminate net, considering all metabolite information in an experiment to examine multiple sets of profiles without bias. There are a myriad of methods for performing untargeted metabolomics.
Globally, liquid chromatography followed by mass spectrometry (LC/MS) is one of the more popular options, providing detection of thousands of peaks in a given biological sample. Each peak is known as a metabolite feature and corresponds to a detected ion with a unique mass-to-charge ratio and retention time, though some metabolites can have more than one peak associated with them.
Untargeted metabolomics samples can include analysis of files that climb into the gigabytes, making manual inspection of the data increasingly impossible. This is when specialized analytical software becomes necessary.
Who Uses Untargeted Metabolomics?
Obviously, targeted and untargeted metabolomics have different applications. But given that untargeted metabolomics is a relatively newer technique, who uses it and how?
Given the breadth of its scope, untargeted metabolomics has a myriad of uses across systems biology, especially since it has already improved our understanding of comprehensive cellular metabolism. For instance, biomedical research has greatly benefited from untargeted metabolomics, proving useful in identifying altered metabolic pathways in disease that represent novel drug targets.
Metabolites from chemical compounds, whether inherent or pharmaceutical, are formed as part of the natural biochemical process of degrading and eliminating the compounds. The rate of degradation of a compound is an important determinant of the duration and intensity of its action. Profiling metabolites of pharmaceutical compounds, drug metabolism, is an important part of drug discovery, leading to an understanding of any undesirable side effects.

Figure 7. Metabolomics is used in drug discovery
Another major use is the characterization of gene and protein functions. Not only does untargeted metabolomics help identify unknown genes and proteins, but also previously unknown functions of known genes and proteins. By screening for metabolites that accumulate after gene mutation or enzyme inhibition, scientists can categorize unanticipated connections between the proteome and the metabolome.